CTRIMS
CTRIMS
Project Information
- Category: Clinical Trial Management System
- Client: MRCG at LSHTM
- Project date: 2024
- Project URL: www.example.com
Project Overview
The Clinical Trial Management System (CTMS) at the Medical Research Council Unit The Gambia at the London School of Hygiene & Tropical Medicine (MRCG at LSHTM) is a digital platform designed to support the planning, execution, monitoring, and oversight of clinical trials.
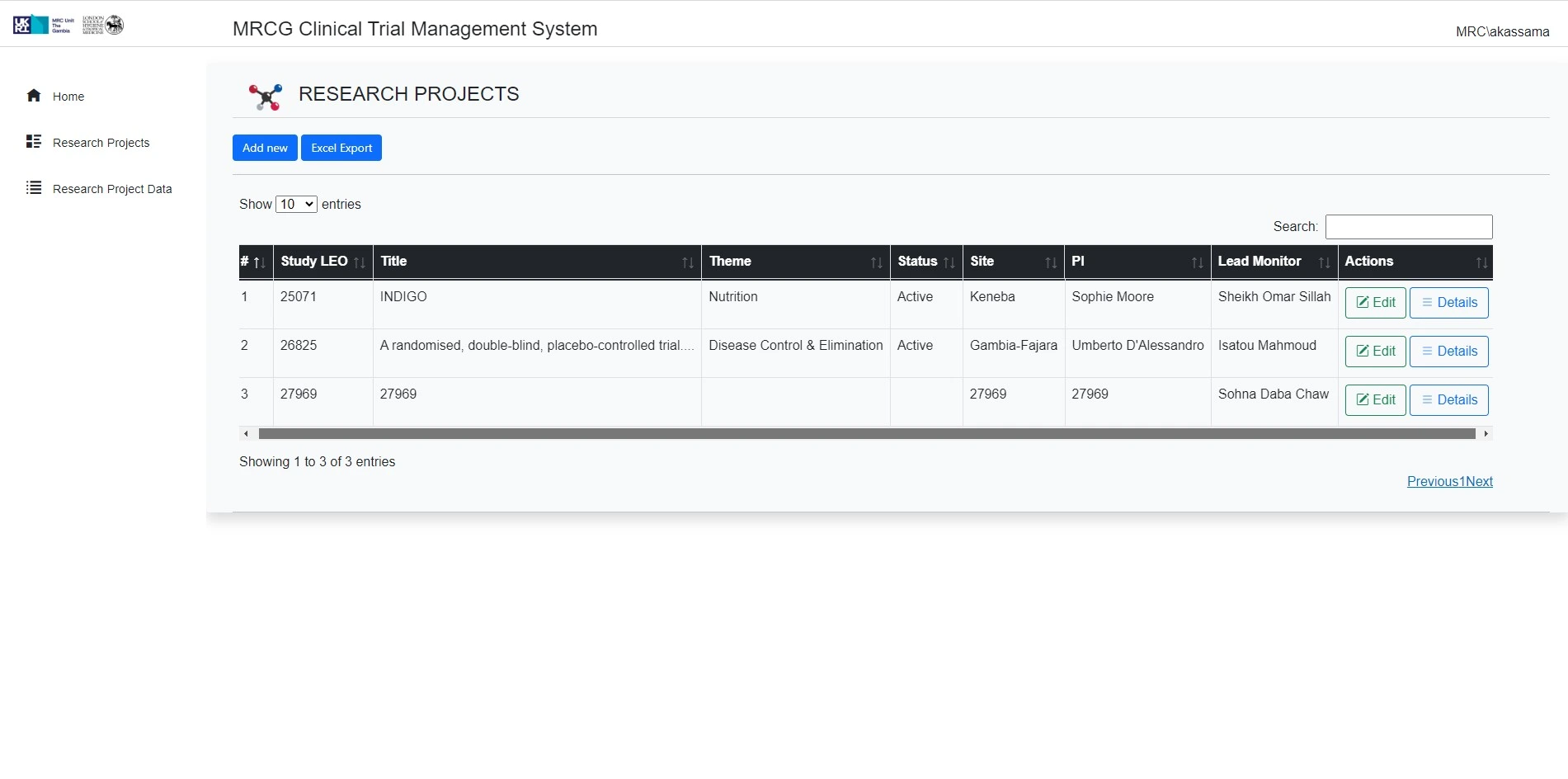
As a clinical trial management system (CTMS), CTRIMS is a critical tool for the MRCG's Clinical Trials Unit. It centralizes and streamlines the complex processes involved in clinical research, from study planning to closeout. The system helps manage subject screening, enrollment, and follow-up, and ensures regulatory adherence and proper data management. This platform supports the CTU in its dual role as an Academic Research Organisation (ARO) and a Contract Research Organisation (CRO), facilitating the effective and compliant execution of clinical trials across West Africa.
Key Features
Centralized management of clinical trial data
The system centralizes and streamlines the complex processes involved in clinical research, from study planning to closeout.
Participant tracking and management
The system helps manage subject screening, enrollment, and follow-up, ensuring regulatory adherence and proper data management.
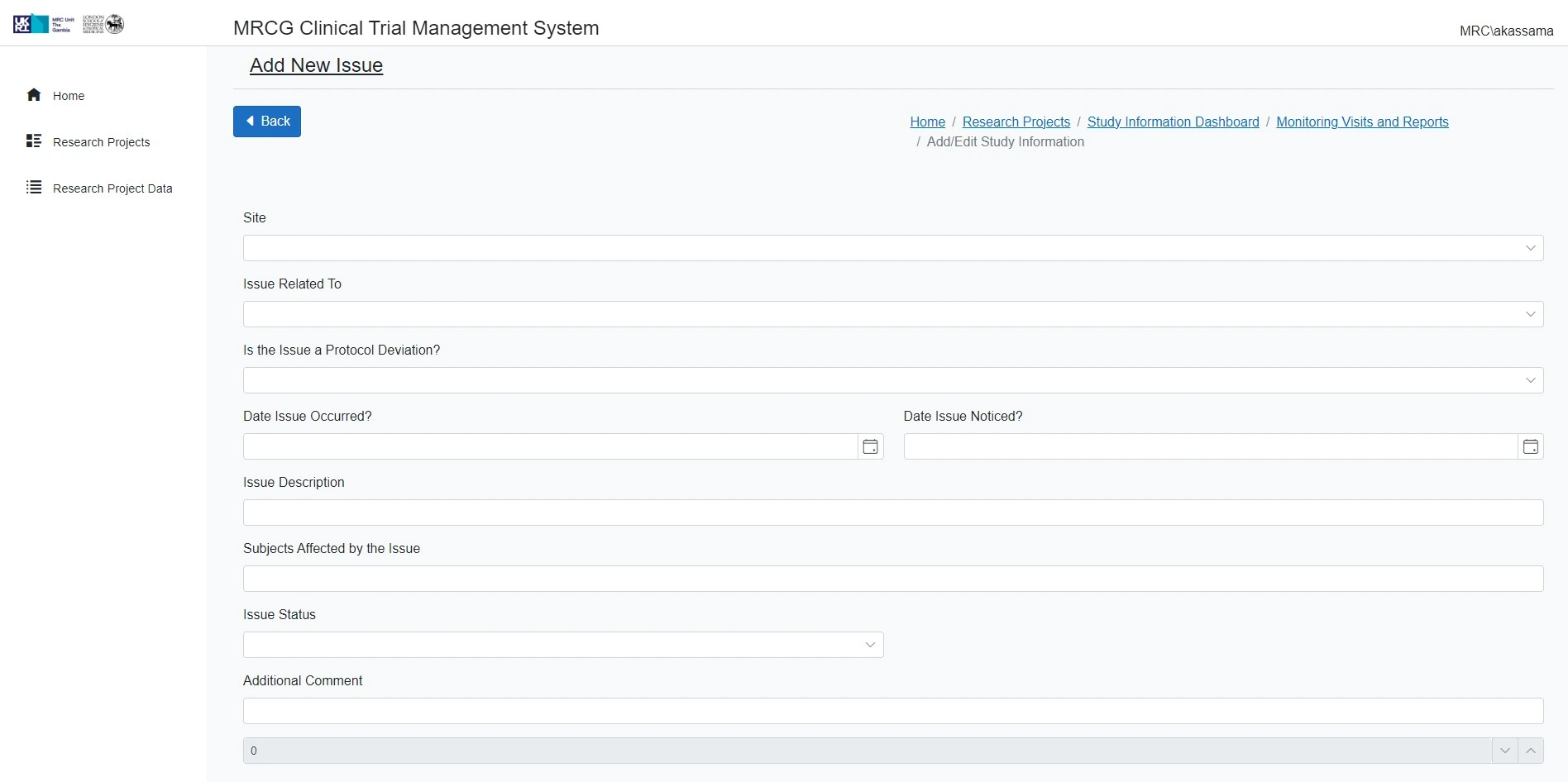
Ensures compliance with Good Clinical Practice (GCP)
This platform supports the CTU in its dual role as an Academic Research Organisation (ARO) and a Contract Research Organisation (CRO), facilitating the effective and compliant execution of clinical trials across West Africa.
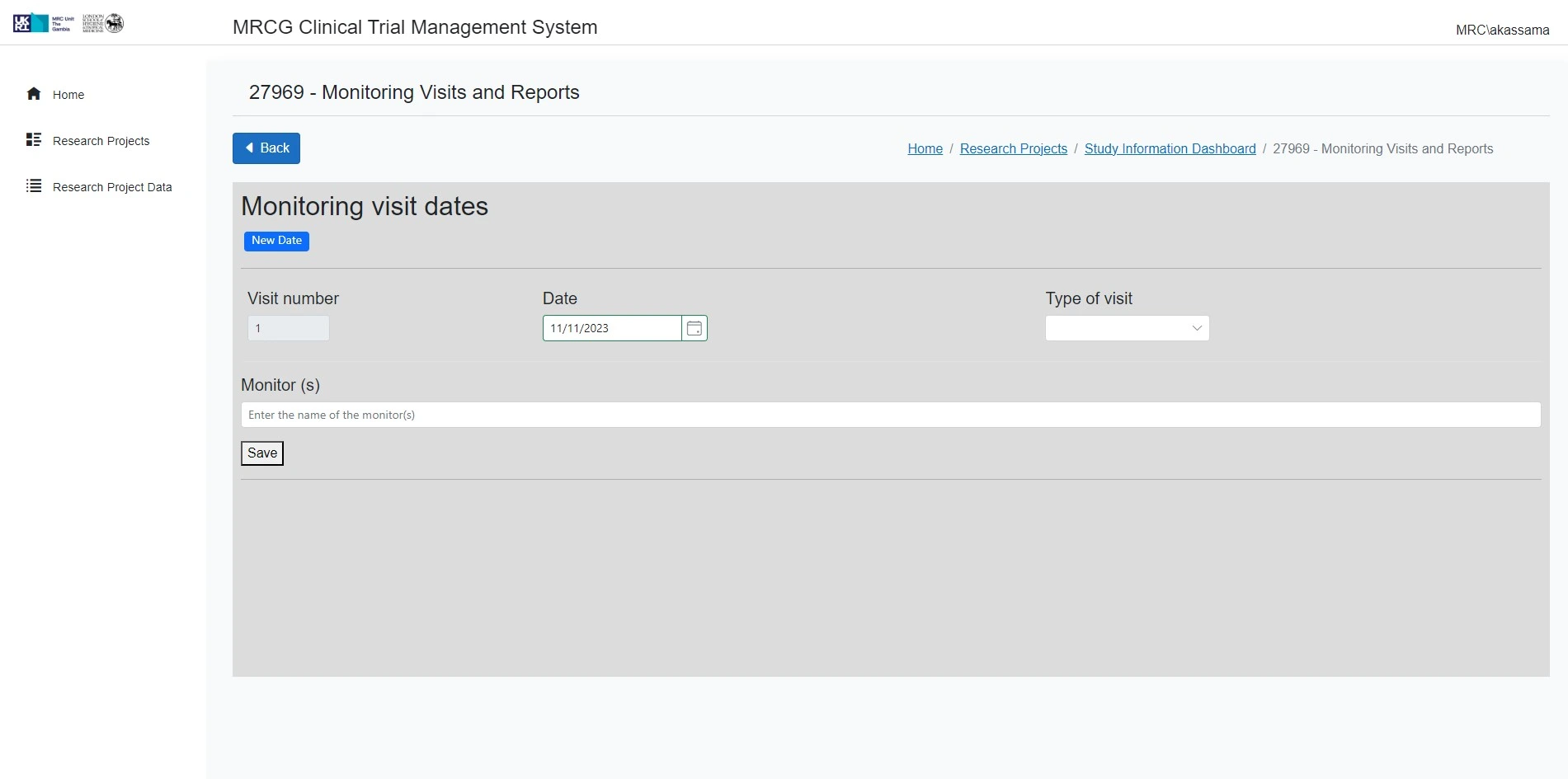
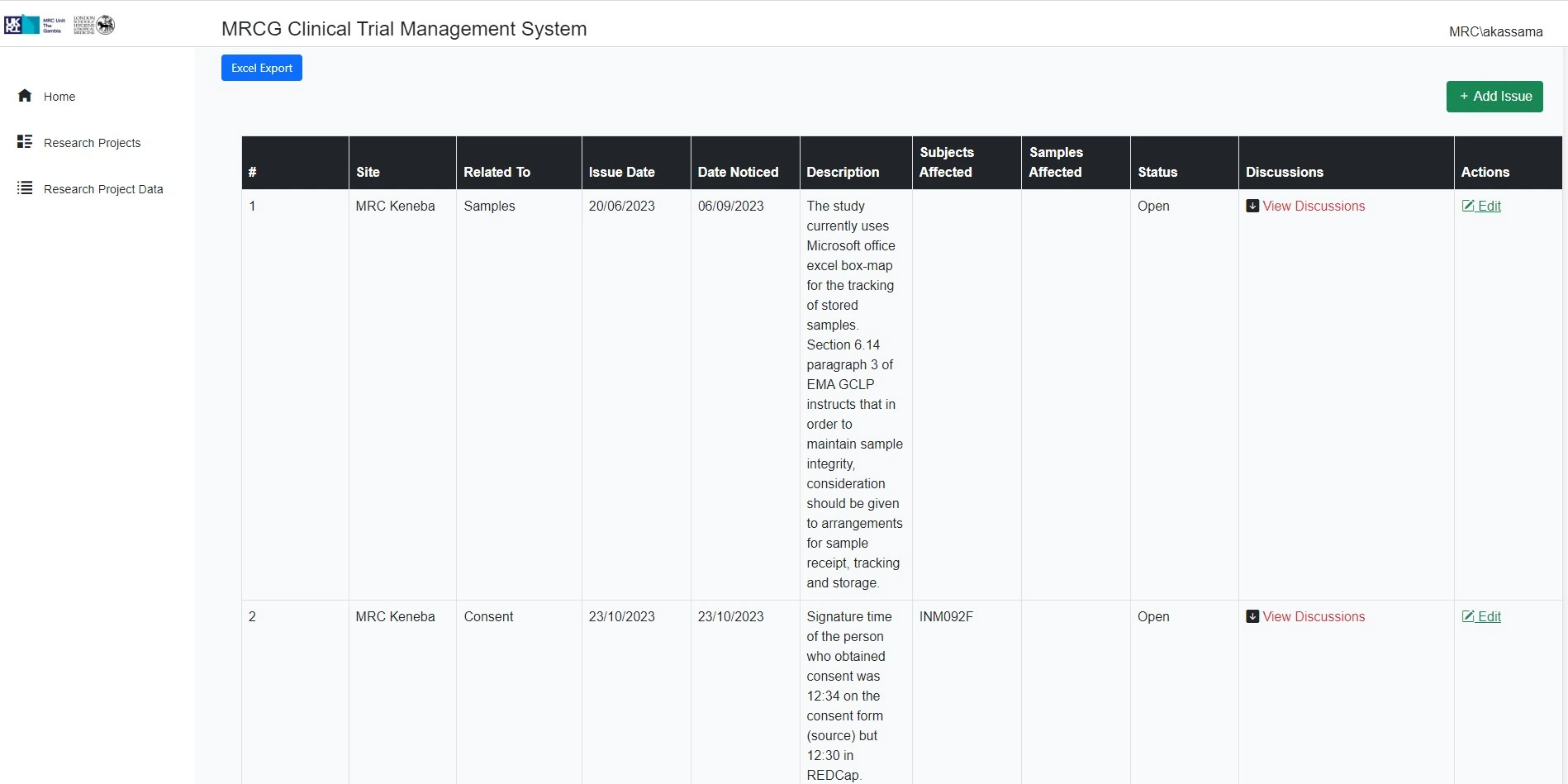
Supports trial oversight and monitoring
The platform ensures a high degree of oversight and monitoring of clinical trials.