CT-UDB
CT-UDB Clinical Trials System
Project Information
- Category: Clinical Trial Utility Database
- Client: MRCG at LSHTM
- Project date: 2024
- Project URL: Internal System
Project Overview
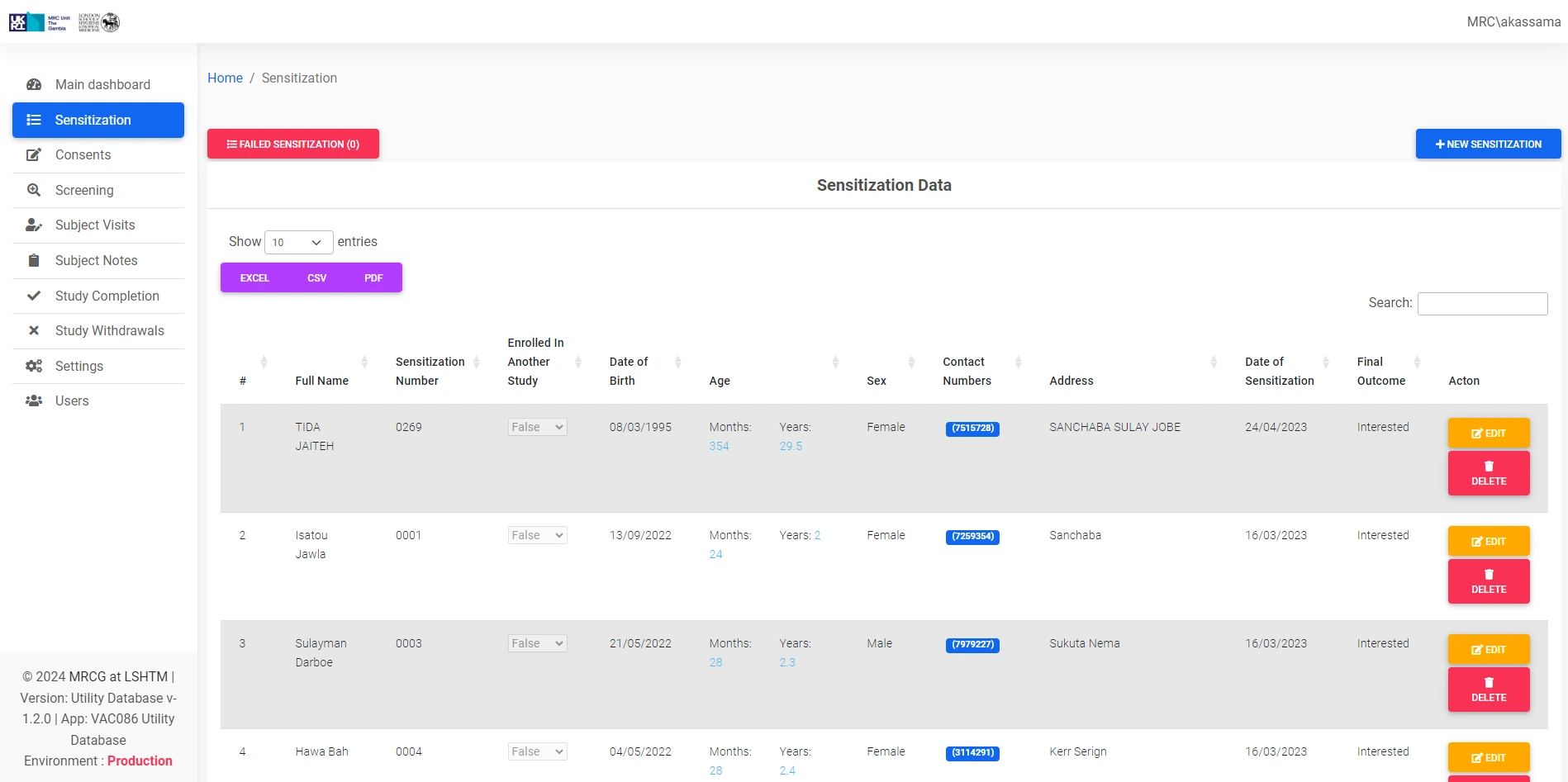
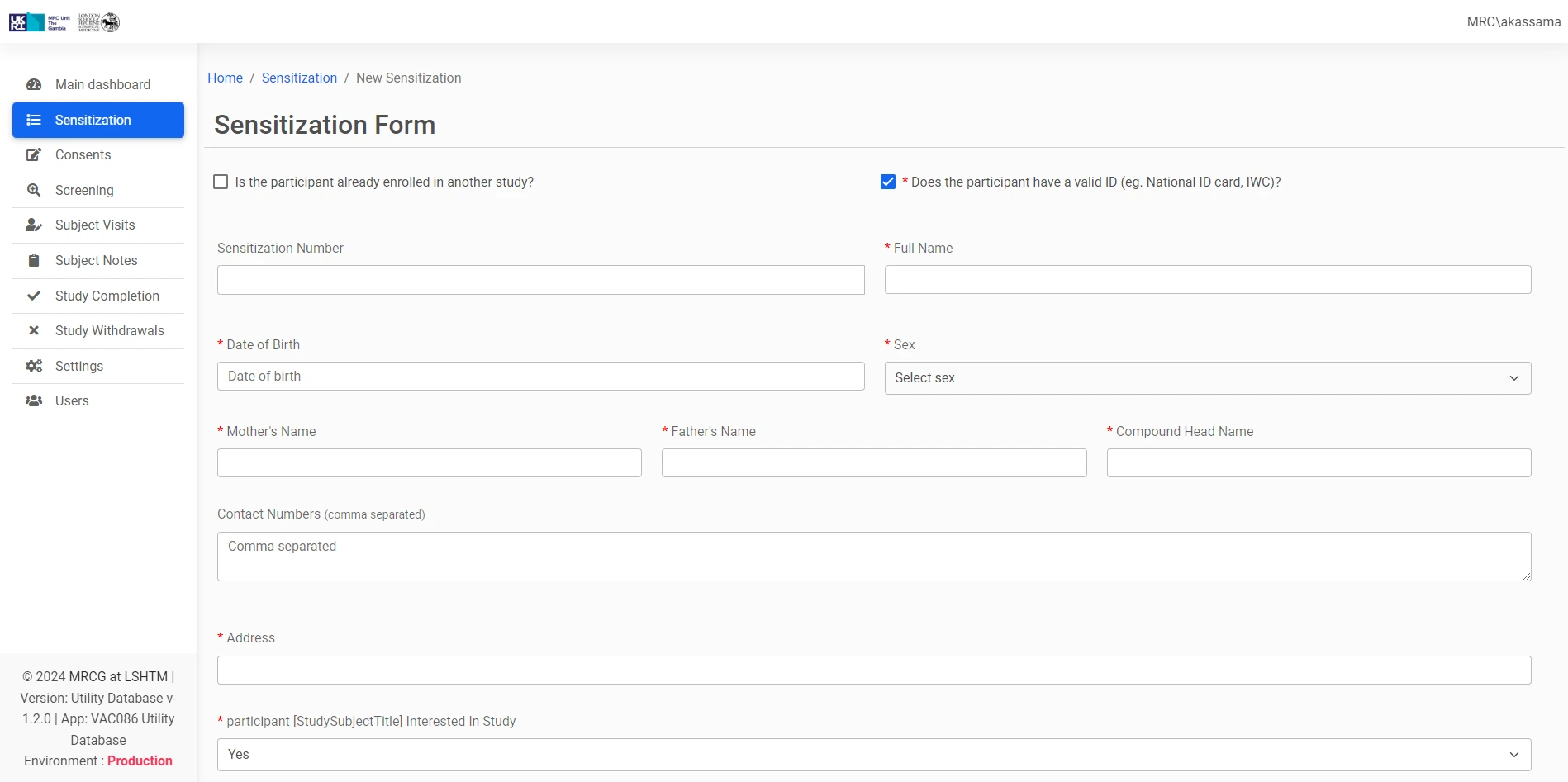
The CT-UDB (Clinical Trials Utility Database) is a digital system developed for the Medical Research Council Unit The Gambia at the London School of Hygiene & Tropical Medicine (MRCG at LSHTM), designed to streamline key operational aspects of clinical trial management and participant engagement.
This specialized utility database complements existing clinical trial systems by providing targeted functionality for specific processes. It serves as a focused tool within the MRCG's secure internal systems ecosystem, supporting pivotal clinical operations and enhancing efficiency and effectiveness in trial management and participant interaction workflows.
Key Features
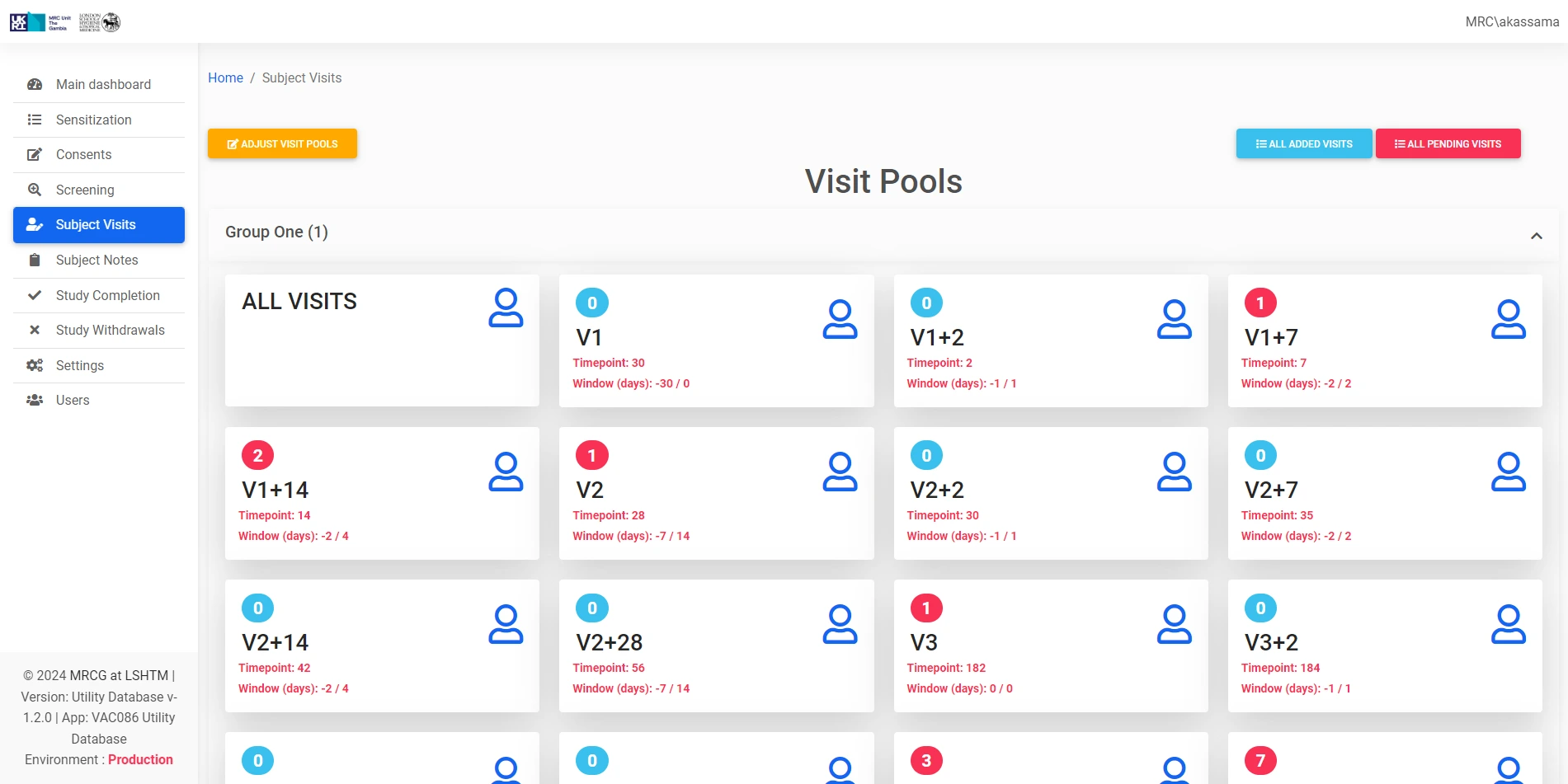
Process Streamlining
Optimizes specific clinical trial workflows and operational processes to improve efficiency and reduce administrative burden on research staff.
Participant Engagement
Enhances management of participant interactions, communication, and engagement throughout the clinical trial lifecycle.
Operational Support
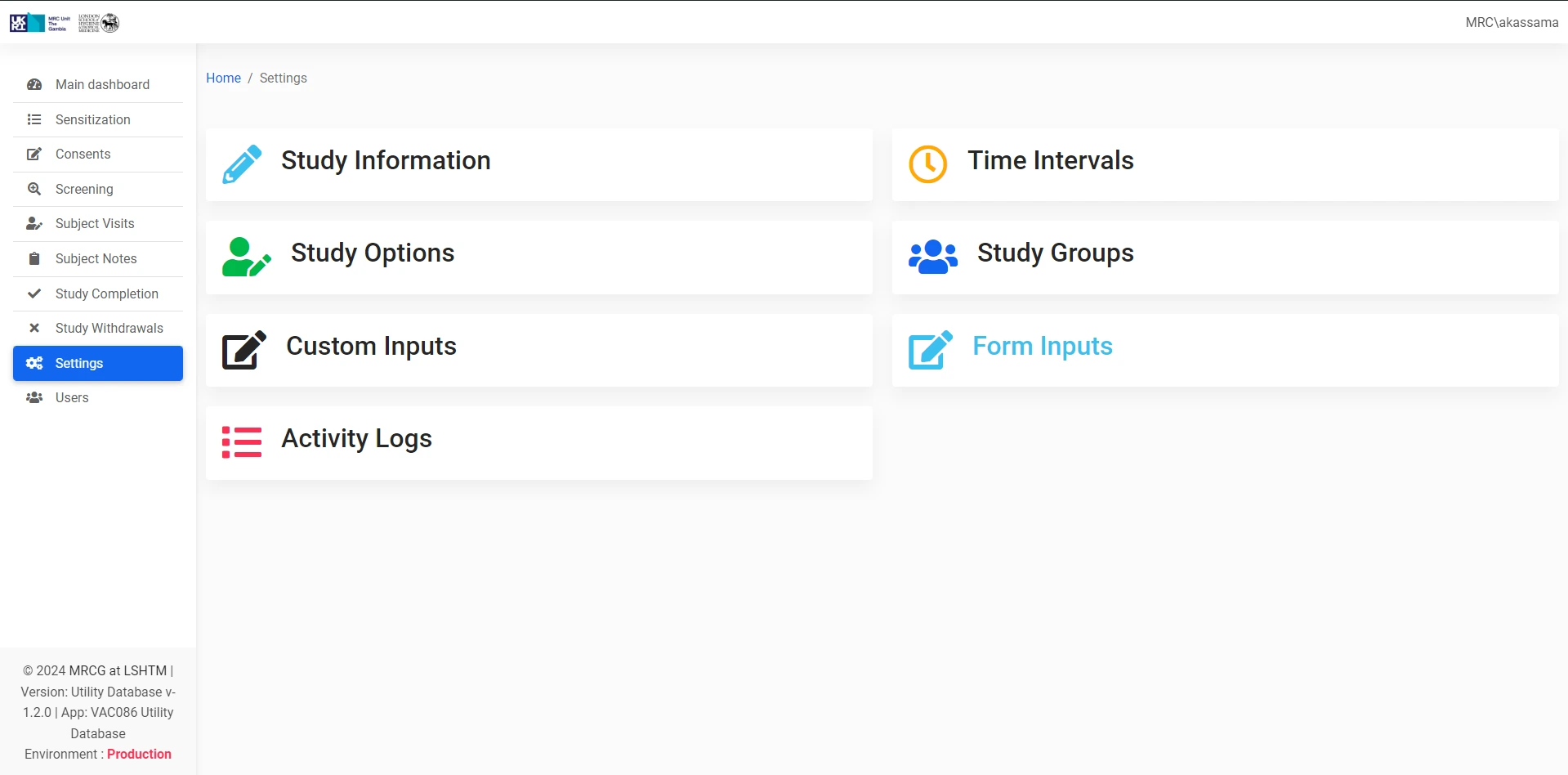
Provides targeted support for key operational functions within the Clinical Trials Unit, complementing existing trial management systems.
Secure Integration
Seamlessly integrates into MRCG's secure internal systems environment, maintaining data integrity and compliance with research protocols.
Targeted Functionality
Focuses on specific data and workflows that support pivotal clinical operations without duplicating existing system capabilities.
Workflow Efficiency
Designed to improve effectiveness in trial management through optimized processes and reduced manual intervention requirements.